
Start your One-Week Free Trial
Already subscribed? Log in »
Heart Tube Looping
Here we will learn how the heart tube elongates and loops to bring its chambers into alignment for blood pumping; once alignment is established, further development of the chambers, outflow tracts, and valves can occur.
We will illustrate the changes that occur by days 22, 23, 24, and 28; bear in mind that these days are approximations, and that intertextual and interindividual variation exist.
The primitive heart tube comprises, from caudal to cranial:
The right and left horns of the sinus venosus.
The primitive atrium and ventricle; label the atrioventricular canal, which is the constricted region between the developing atria and ventricle.
And, the bulbus cordis and truncus arteriosus, which comprise the outflow tract.
The aortic sac is at the cranial-most end; this will be the first part of the aorta to develop.
Blood is pumped through the outflow tract by day 22.
On day 23, progenitor cells from the second heart field (SHF) are added to the tube; as it elongates, the tube changes shape.
Elongation bends the tube ventrally and rightward (dextral looping), producing a C-shaped loop.
Looping and elongating ruptures the dorsal mesocardium, which anchored the heart to the dorsal body wall; the rupture creates the transverse sinus of the pericardium.
By day 24, further elongation and bending creates an S-shape.
Growth displaces the ventricle ventrally, caudally, and to the left, and the bulbous cordis shifts ventrally, caudally, and to the right; the atrium is pushed dorsally and cranially.
By this stage, blood circulates throughout the embryo.
By day 28, elongation is complete, and the tube approximates a U-shape, so that the atria and ventricles are properly aligned for blood flow.
Notice that the truncus arteriosus is continuous cranially with the aortic sac, which as we’ll learn elsewhere, gives rise to the aortic arches.
The bulbus cordis expanded caudally and can be subdivided into two parts:
The conus cordis, which becomes part of the outflow tract, and the developing primitive right ventricle.
We highlight the primitive left ventricle and the bulboventricular sulcus, which is the external indentation that marks the internal primary muscular fold (formerly called the bulboventricular fold).
The sinus venosus shifts dorsally; as we’ll learn elsewhere, it undergoes remodeling to form, most notably, portions of the right atrium and the orifices of the caval veins and coronary sinus.
These conotruncal anomalies arise from malformations of the outflow tract and great vessels.
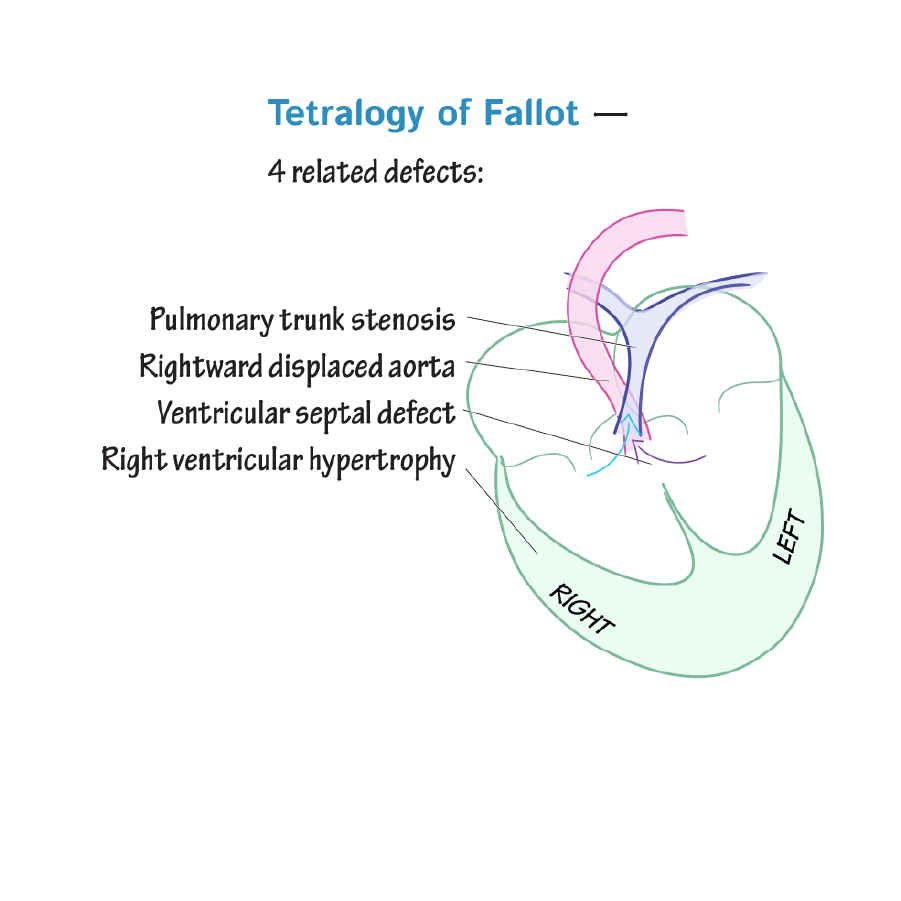
Tetralogy of Fallot is characterized by four defects: pulmonary stenosis, ventricular septal defect, overriding aorta, and hypertrophy of the right ventricle.
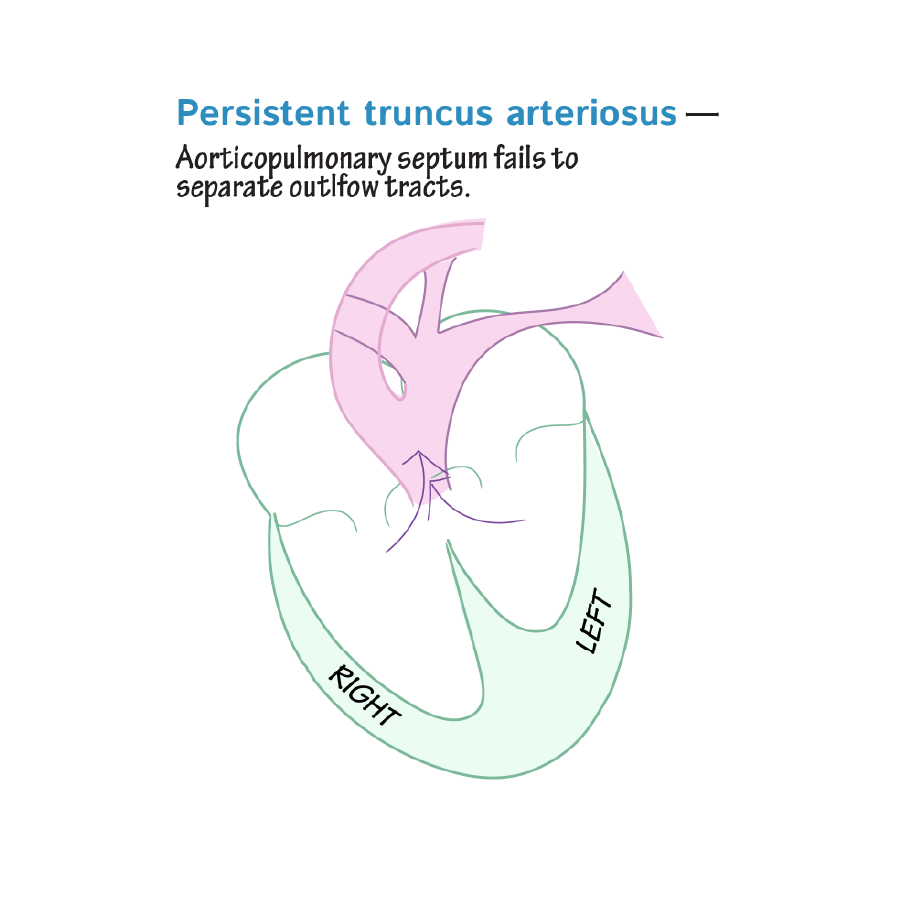
 Persistent truncus arteriosus is due to failure of the truncus arteriosus to divide into the aorta and pulmonary trunk; thus, mixed blood is sent to the pulmonary and systemic circulatory systems.
Persistent truncus arteriosus is due to failure of the truncus arteriosus to divide into the aorta and pulmonary trunk; thus, mixed blood is sent to the pulmonary and systemic circulatory systems.
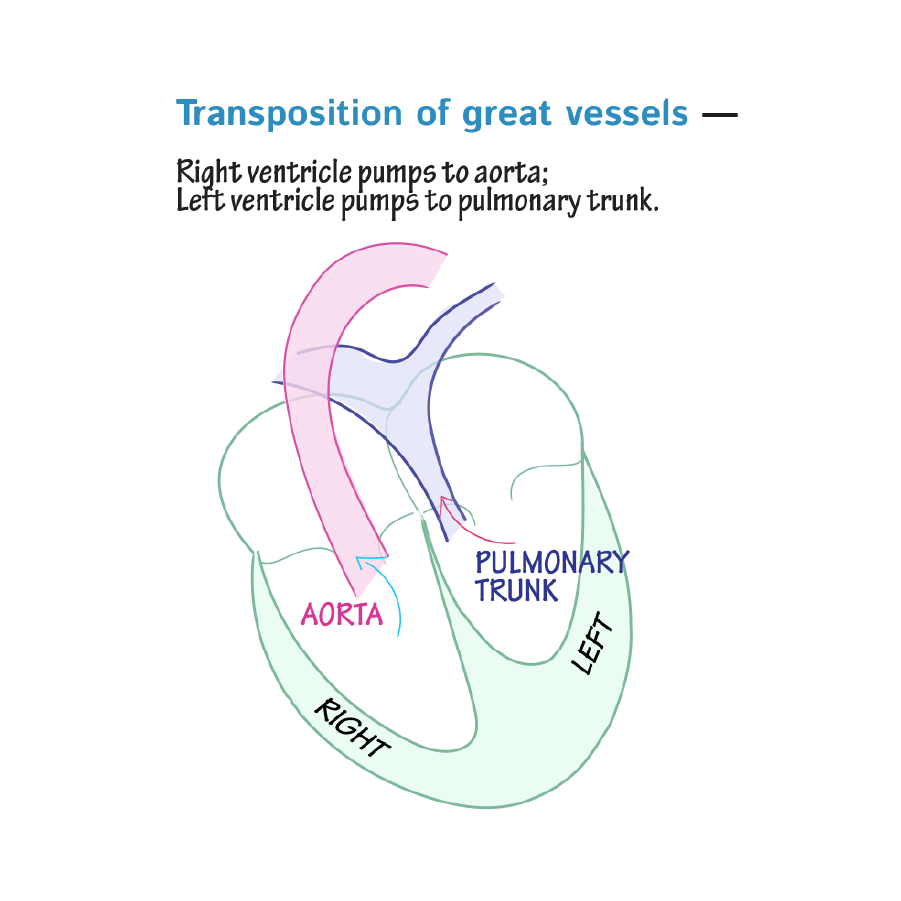
 Transposition of the great arteries occurs when the pulmonary artery and aorta are switched in position, which causes oxygen-poor blood to be sent to the systemic circulation and oxygen-rich blood to be sent to the pulmonary circulation.
Transposition of the great arteries occurs when the pulmonary artery and aorta are switched in position, which causes oxygen-poor blood to be sent to the systemic circulation and oxygen-rich blood to be sent to the pulmonary circulation.
 Lastly, double-outlet right ventricle occurs when both the aorta and the pulmonary artery arise from the right ventricle, so oxygen-poor blood is sent to the body. The condition of double-outlet right ventricle is often associated with ventricular septal defect, in which a hole in the septum allows oxygen-rich blood to move from the ventricle to the aorta and pulmonary artery.
Lastly, double-outlet right ventricle occurs when both the aorta and the pulmonary artery arise from the right ventricle, so oxygen-poor blood is sent to the body. The condition of double-outlet right ventricle is often associated with ventricular septal defect, in which a hole in the septum allows oxygen-rich blood to move from the ventricle to the aorta and pulmonary artery.
Day 22
Day 23
Day 24
Day 28
Conotruncal Anomalies
 Persistent truncus arteriosus is due to failure of the truncus arteriosus to divide into the aorta and pulmonary trunk; thus, mixed blood is sent to the pulmonary and systemic circulatory systems.
Persistent truncus arteriosus is due to failure of the truncus arteriosus to divide into the aorta and pulmonary trunk; thus, mixed blood is sent to the pulmonary and systemic circulatory systems.
 Transposition of the great arteries occurs when the pulmonary artery and aorta are switched in position, which causes oxygen-poor blood to be sent to the systemic circulation and oxygen-rich blood to be sent to the pulmonary circulation.
Transposition of the great arteries occurs when the pulmonary artery and aorta are switched in position, which causes oxygen-poor blood to be sent to the systemic circulation and oxygen-rich blood to be sent to the pulmonary circulation.
 Lastly, double-outlet right ventricle occurs when both the aorta and the pulmonary artery arise from the right ventricle, so oxygen-poor blood is sent to the body. The condition of double-outlet right ventricle is often associated with ventricular septal defect, in which a hole in the septum allows oxygen-rich blood to move from the ventricle to the aorta and pulmonary artery.
Lastly, double-outlet right ventricle occurs when both the aorta and the pulmonary artery arise from the right ventricle, so oxygen-poor blood is sent to the body. The condition of double-outlet right ventricle is often associated with ventricular septal defect, in which a hole in the septum allows oxygen-rich blood to move from the ventricle to the aorta and pulmonary artery.



