Start your One-Week Free Trial
Already subscribed? Log in »
Elimination Kinetics
Elimination Kinetics
Overview
- Elimination kinetics is the study of the rate at which drugs clear from the body in relation to their plasma concentration.
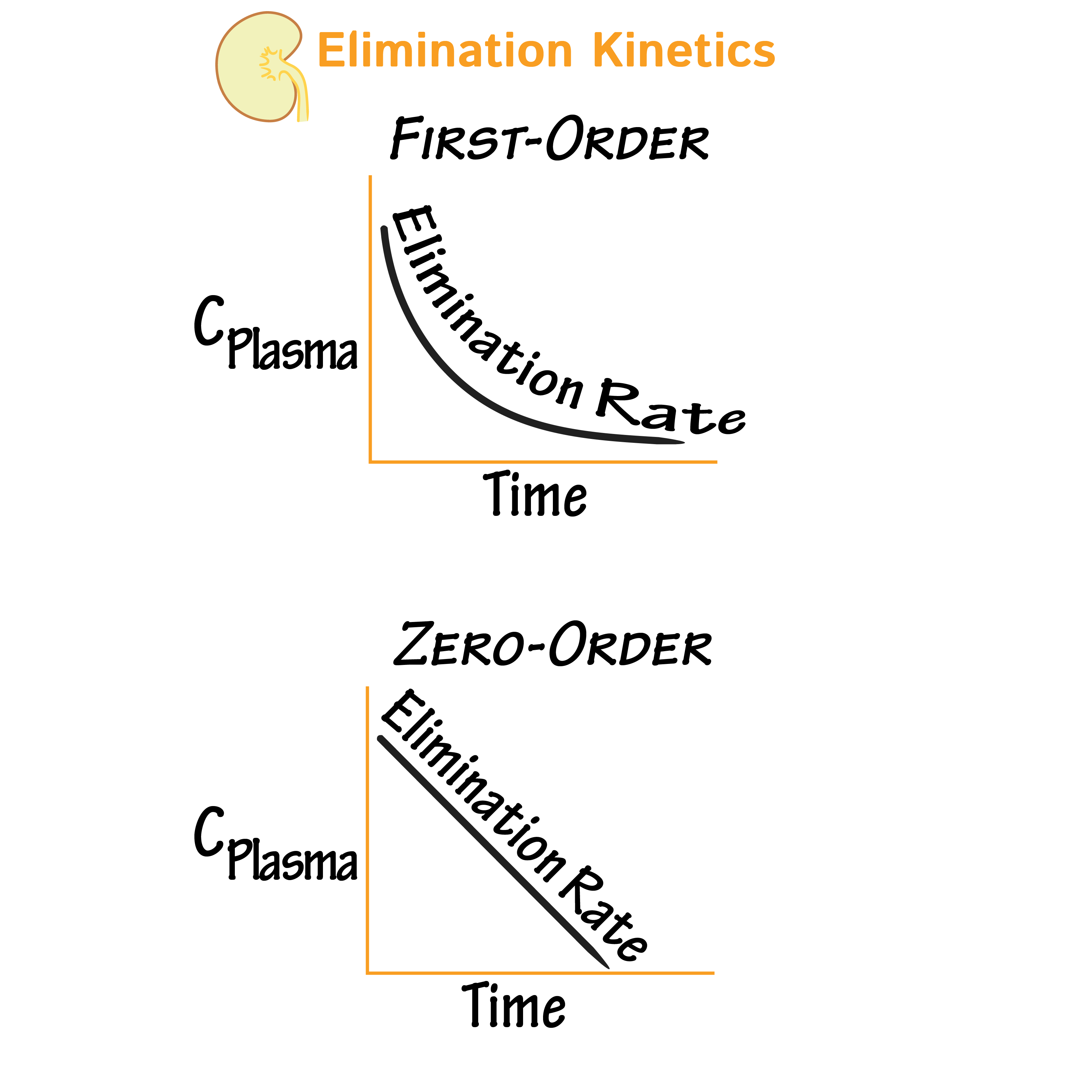
First-Order Elimination
- In first-order kinetics, the rate of elimination is proportional to the concentration of the drug: the higher the concentration, the greater the relative elimination rate.
- The elimination rate curve is exponential; the rate of elimination decreases in exponentially with time.
Example
- Assume, for instance that the half life of a drug is 1 hour, meaning 50% of the drug is eliminated every hour, and the starting concentration is 20.
- Time 0: Elimination Rate is 10 units/hr
- Time 1hr: Elimination Rate is 5 units/hr
- Time 3hrs: Elimination Rate is 2.5 units/hr
- Time 4hrs: Elimination Rate is 1.25 units/hr
Zero-Order Elimination
- In zero-order kinetics, the rate of elimination is constant regardless of concentration.
- This occurs when a drug saturates its elimination mechanisms, so regardless of the reduction in concentration, the rate of elimination remains unchanged.
- The elimination rate curve is linear; the rate of elimination decreases is a constant, linear fashion.
Example
- If the concentration was 20 and 5 units were removed every hour, then in 1 hour, the concentration would be 15; in 2 hours it would be 10; in 3 hours it would be 5.
- Key examples of drugs with zero-order kinetics are phenytoin, ethanol, and aspirin.
- As well, if it is a controlled release medication or a medication that is rate limited by gastric emptying, the drug may demonstrate zero-order kinetics.
